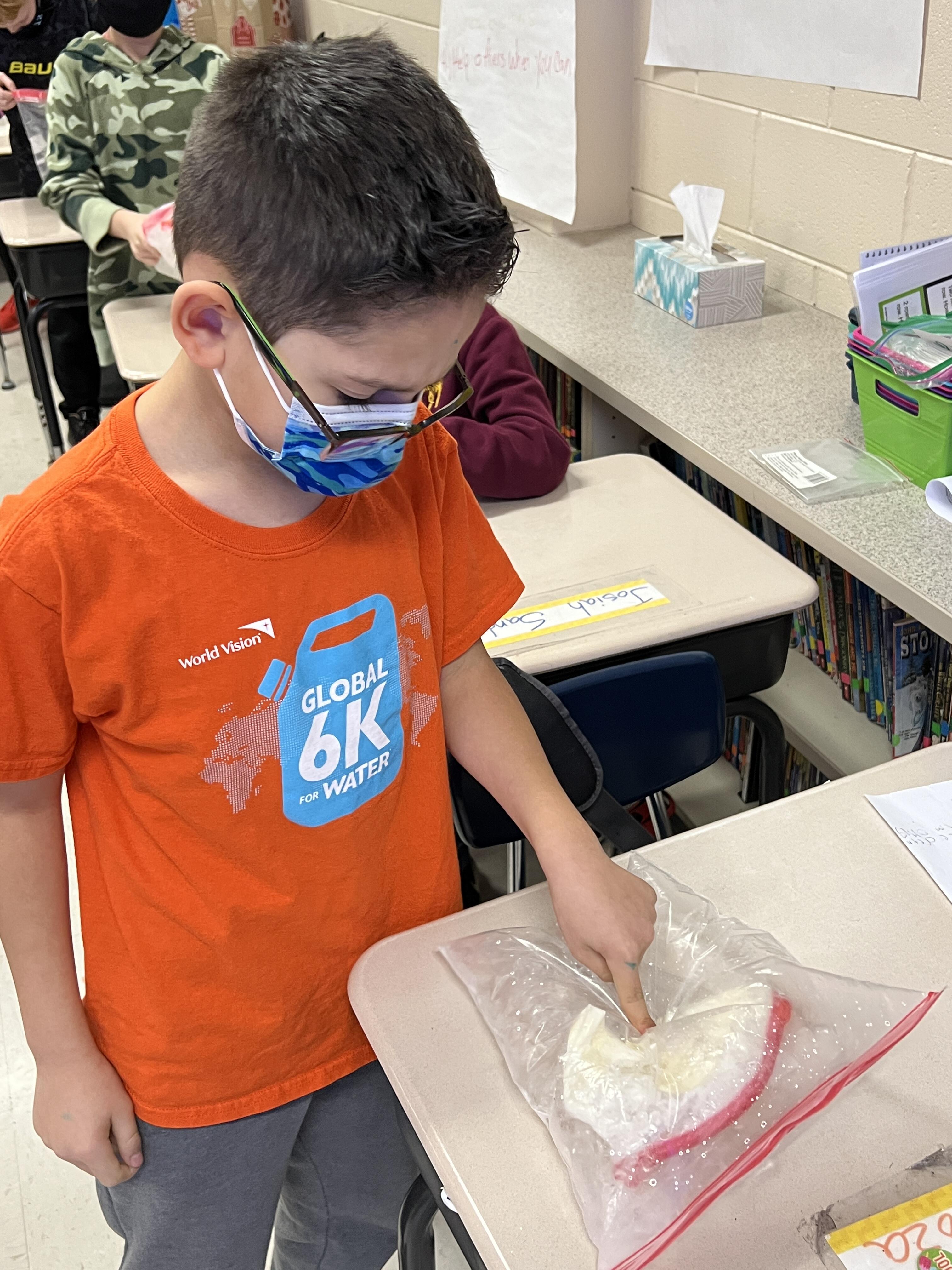
At the culmination of their Energy Conversions unit in Science, Mrs. Duignan's 4th grade class learned about making ice cream. Ms. Stephens, Sward's ELL teacher invited a special guest to join Mrs. Duignan's class for this exciting experiment. Ms. Stephens' father is an engineer who visited Mrs. Duignan's class to help students learn about how engineers are problem solvers. The students were tasked to act like engineers by solving the problem of how to transfer the heat out of the cream and sugar to make ice-cream. The goal of the lesson was to explain how heat energy transfers through conduction (solids), convection (gas and liquids), and radiation (air and space).
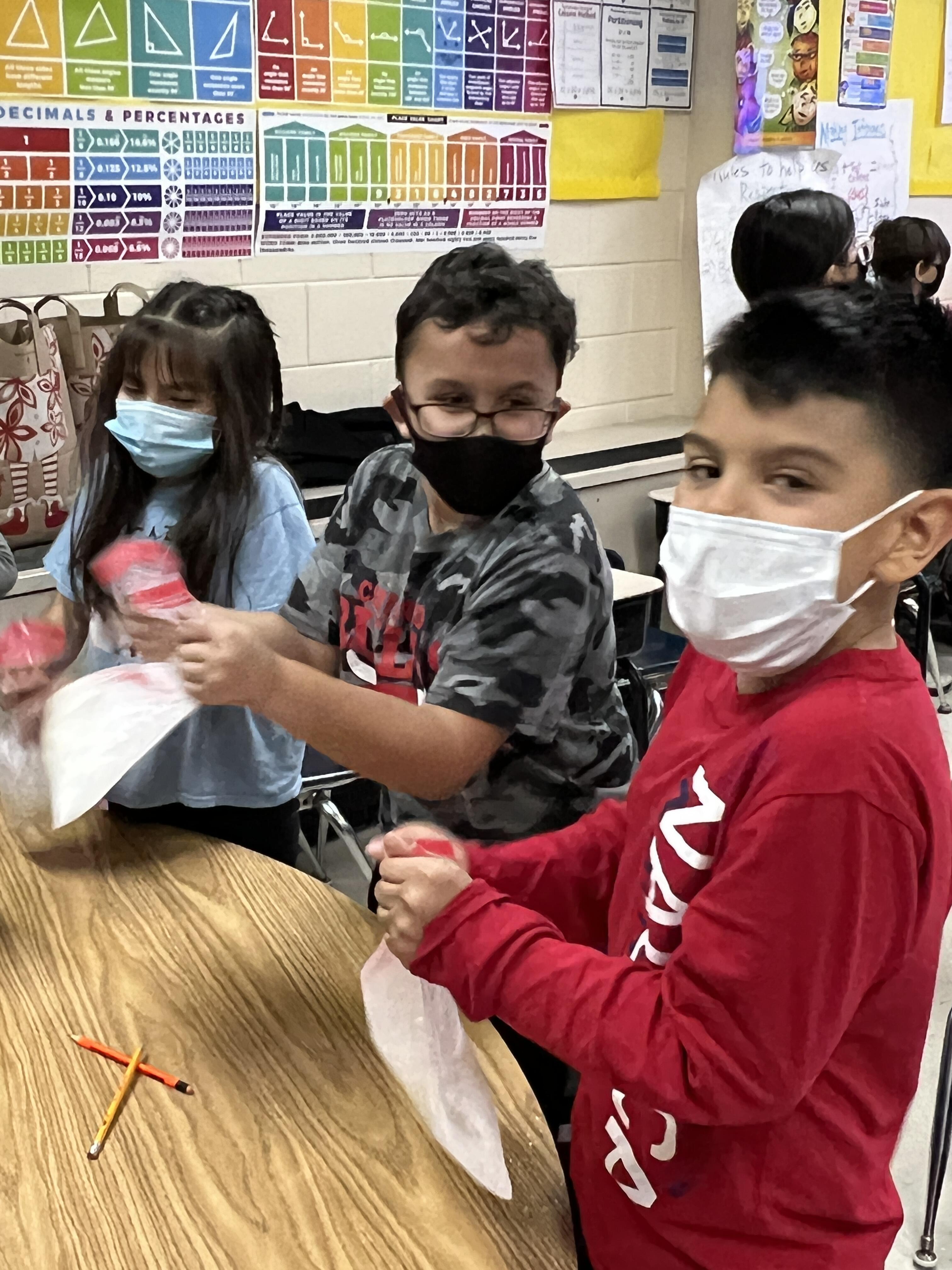
The students learned how removing heat from the milk would turn it into ice cream. When the heat is traveling through the liquid milk that is the process of convection. When the heat is traveling through the solid ice that is conduction. Radiation is happening all the time through the air. That was the application of the 3 ways of heat transfer to the ice cream experiment. When the kids were asked about the experiment they said they were surprised at how quickly the ice cream became cold and solid. They didn't understand why we put salt with the ice so we explained that that makes the ice cooler and decreases the freezing point. So even though the ice will melt, the salt will keep it very cold.
Mrs. Duignan's student Nathan said "It was a fun, interesting and delicious way to learn." When asked about the most challenging part of the experiment, Nathan commented "when you would shake it, and the bag would leak, it would get everywhere. Your arms start to hurt when you shake it for awhile." Overall, Mrs. Duignan and Ms. Stephens reported that the students really enjoyed the lesson and that they definitely have a solid understanding of heat transfer after this super COOL experiment!